 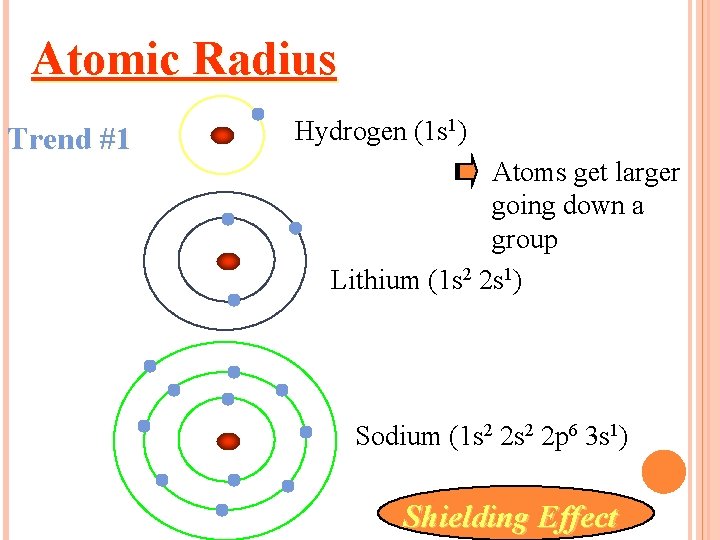
This is because although the valence shell maintains the same principal quantum number, the number of protons-and hence the nuclear charge-is increasing as you go across the row. Going across a row on the periodic table, left to right, the trend is different. 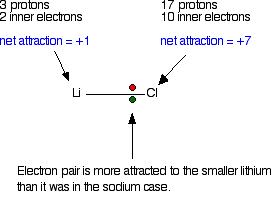
This trend can be summarized as follows: as ↓ PT, atomic radius ↑ This is because the valence electron shell is getting a larger and there is a larger principal quantum number, so the valence shell lies physically farther away from the nucleus. Such radii can be estimated from various experimental techniques, such as the x-ray crystallography of crystals.Īs you go down a column of the periodic table, the atomic radii increase. Although the concept of a definite radius of an atom is a bit fuzzy, atoms behave as if they have a certain radius. The atomic radius An indication of the size of the atom. The first periodic trend we will consider atomic radius. 
There may be a few points where an opposite trend is seen, but there is an overall trend when considered across a whole row or down a whole column of the periodic table. There is no other tool in science that allows us to judge relative properties of a class of objects like this, which makes the periodic table a very useful tool. The variation of properties versus position on the periodic table is called periodic trends Variation of properties versus position on the periodic table. 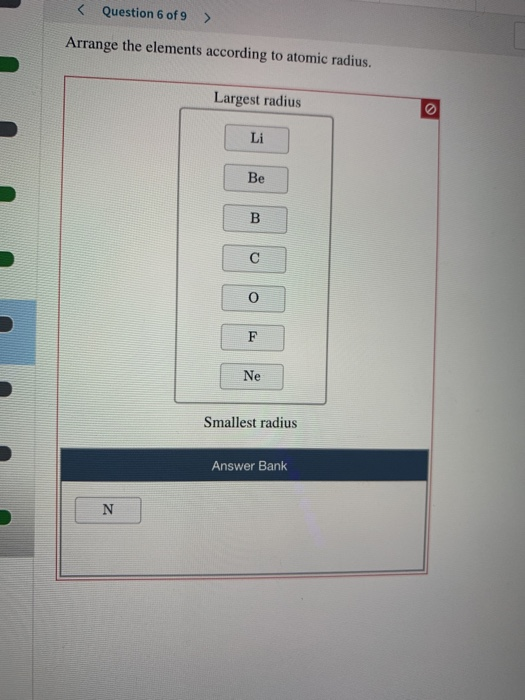
One of the reasons the periodic table is so useful is because its structure allows us to qualitatively determine how some properties of the elements vary versus their position on the periodic table. Be able to state how certain properties of atoms vary based on their relative position on the periodic table.Get the free view of chapter 7 Modern Periodic Table 11th extra questions for Chemistry 11th Standard Maharashtra State Board and can use Shaalaa. Maximum students of Maharashtra State Board 11th prefer Balbharati Textbook Solutions to score more in exam. The questions involved in Balbharati Solutions are important questions Using Balbharati 11th solutions Modern Periodic Table exercise by students are an easy way to prepare for the exams, as they involve solutions arranged chapter-wise also page wise. Balbharati textbook solutions can be a core help for self-study and acts as a perfect self-help guidance for students.Ĭoncepts covered in Chemistry 11th Standard Maharashtra State Board chapter 7 Modern Periodic Table are Introduction of Periodic Table, Structure of the Modern Periodic Table, Periodic Table and Electronic Configuration, Blockwise Characteristics of Elements, Periodic Trends in Elemental Properties. has the Maharashtra State Board Chemistry 11th Standard Maharashtra State Board solutions in a manner that help students grasp basic concepts better and faster.įurther, we at provide such solutions so that students can prepare for written exams. The detailed, step-by-step solutions will help you understand the concepts better and clear your confusions, if any. This will clear students doubts about any question and improve application skills while preparing for board exams. Balbharati solutions for Chemistry 11th Standard Maharashtra State Board chapter 7 (Modern Periodic Table) include all questions with solution and detail explanation. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
